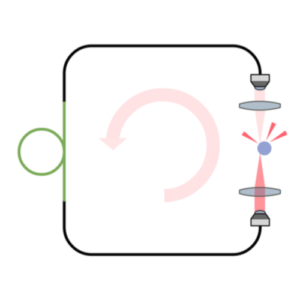
Intracavity optical trapping of microscopic particles in a ring-cavity fiber laser
Fatemeh Kalantarifard, Parviz Elahi, Ghaith Makey, Onofrio M. Maragò, F. Ömer Ilday & Giovanni Volpe
Nature Communications 10, 2683 (2019)
doi: 10.1038/s41467-019-10662-7
arXiv: 1808.07831
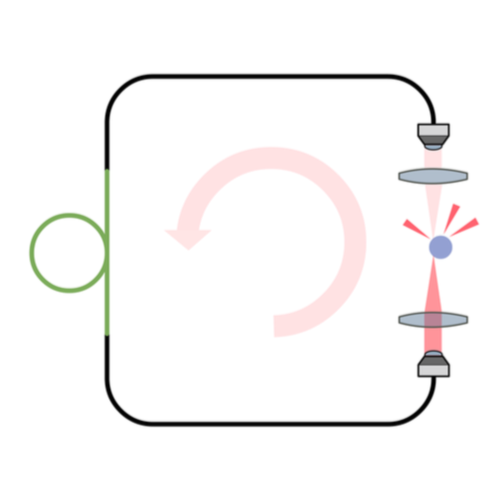
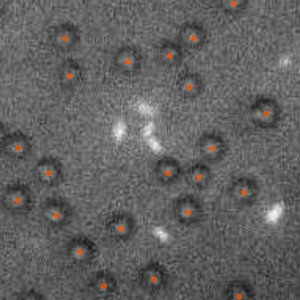
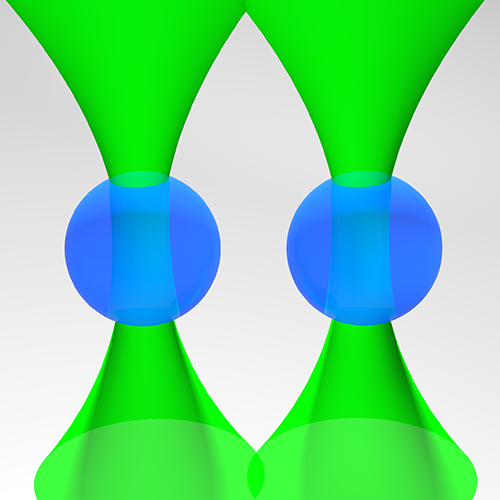
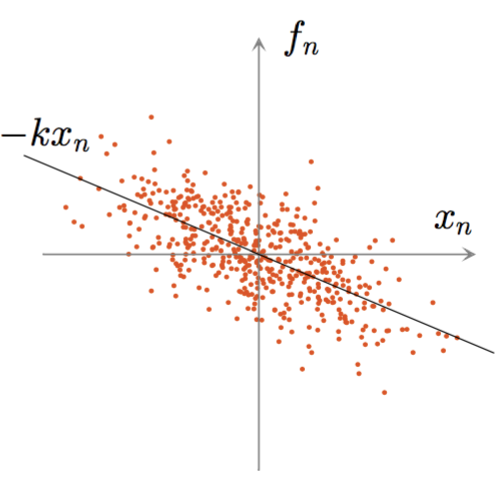
Standard optical tweezers rely on optical forces arising when a focused laser beam interacts with a microscopic particle: scattering forces, pushing the particle along the beam direction, and gradient forces, attracting it towards the high-intensity focal spot. Importantly, the incoming laser beam is not affected by the particle position because the particle is outside the laser cavity. Here, we demonstrate that intracavity nonlinear feedback forces emerge when the particle is placed inside the optical cavity, resulting in orders-of-magnitude higher confinement along the three axes per unit laser intensity on the sample. This scheme allows trapping at very low numerical apertures and reduces the laser intensity to which the particle is exposed by two orders of magnitude compared to a standard 3D optical tweezers. These results are highly relevant for many applications requiring manipulation of samples that are subject to photodamage, such as in biophysics and nanosciences.